Buclizine Hydrochloride
Looking for a reliable supplier of Buclizine Hydrochloride?
Get competitive pricing and technical support from our experts
Premium Quality
Buclizine Hydrochloride
Buclizine Hydrochloride is a first-generation antihistamine with strong appetite-stimulating effects. Produced under WHO-GMP certified facilities, it is manufactured to meet global pharmacopoeial standards, ensuring consistent quality and therapeutic reliability. Widely trusted for its efficacy in managing motion sickness and nausea, it also plays a significant role in nutritional therapy where appetite stimulation is critical.
D.K. Pharmachem ensures every batch is tested for assay, impurities, and stability, providing ≥99% purity with full regulatory documentation (DMF/EDMF). Our proven supply reliability makes us a trusted partner for pharmaceutical companies worldwide.
Product Specifications
| Attribute | Details |
|---|---|
| Pharmacopoeia Standard | USP / EP |
| CAS Number | 2185-92-4 |
| Molecular Formula | C₂₈H₃₃ClN₂ |
| Molecular Weight | 433.03 g/mol |
| Therapeutic Application | Antihistamine & Appetite Stimulant |
| DMF / EDMF Status | Available |
| Manufacturing Standard | WHO-GMP Certified |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99% (HPLC) |
Diverse Applications

Pharmaceutical Formulations
Key active ingredient in anti-nausea and anti-motion sickness tablets and syrups

Appetite Stimulation
Widely used in pediatric and geriatric formulations for undernutrition and recovery support

Global Market Demand
Preferred by generic manufacturers due to proven efficacy and low side-effect profile

Research Institutions
Supports studies in antihistamine receptor activity and orexigenic mechanisms
Other Products
Chloramine Y
CAS: 23239-51-2
Grade: EP/BP

Antimicrobial Agent

WHO-GMP Certified
Chloramine Y
CAS: 23239-51-2
Grade: EP/BP

Antimicrobial Agent

WHO-GMP Certified
Chloramine Y
CAS: 23239-51-2
Grade: EP/BP

Antimicrobial Agent

WHO-GMP Certified
Chloramine Y
CAS: 23239-51-2
Grade: EP/BP

Antimicrobial Agent

WHO-GMP Certified
Chloramine Y
CAS: 23239-51-2
Grade: EP/BP

Antimicrobial Agent

WHO-GMP Certified
Buclizine Hydrochloride API Manufacturer in India
WHO-GMP certified bulk Buclizine HCl supplier for global pharmaceutical formulators
Looking for a reliable supplier of Buclizine Hydrochloride?
Premium Quality Buclizine Hydrochloride
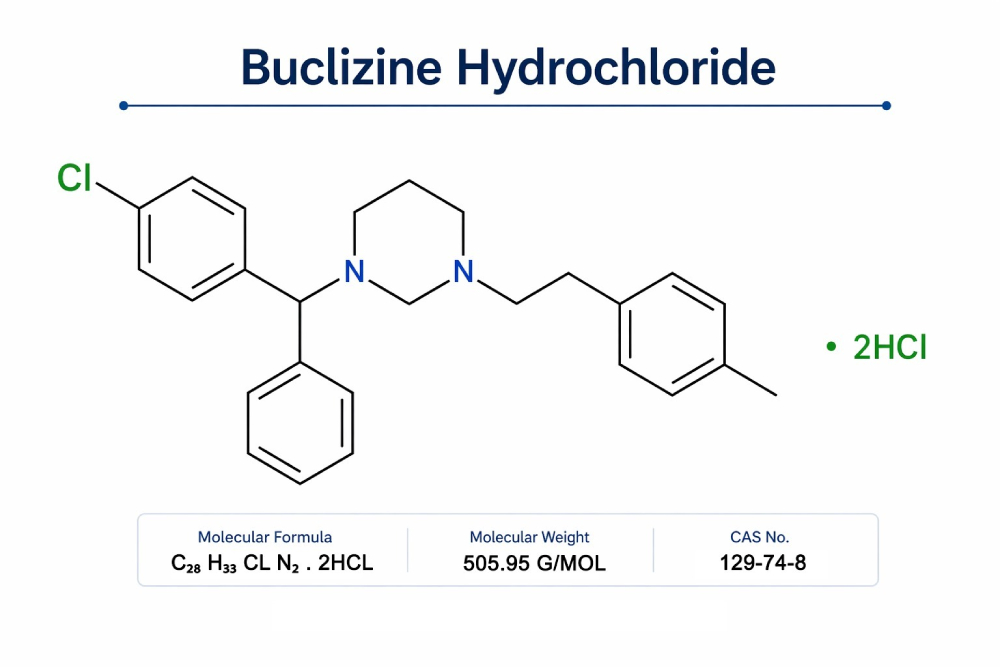
D.K. Pharmachem Pvt. Ltd. is among India’s trusted buclizine hydrochloride API manufacturers, supplying high-quality buclizine HCl for pharmaceutical formulations. Buclizine Hydrochloride is widely used as an antiemetic API, commonly formulated into buclizine hydrochloride tablets for the treatment of nausea, vomiting and motion sickness.
With decades of experience in API manufacturing, we cater to domestic and international markets with consistent quality, regulatory compliance and reliable bulk supply.
Buclizine Hydrochloride API
Technical Specifications
| Attribute | Details |
|---|---|
| Pharmacopoeia Standard | USP / EP |
| CAS Number | 129-74-8 |
| Molecular Formula | C₂₈ H₃₃ Cl N₂ . 2HCl |
| Molecular Weight | 505.95 g/mol |
| Therapeutic Application | Antihistamine & Antiemetic |
| DMF / EDMF Status | Available |
| Manufacturing Standard | WHO-GMP Certified |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99% (HPLC) |
Manufacturing Capabilities & Supply Strength
Buclizine Hydrochloride API at DK Pharmachem is manufactured through a controlled chemical synthesis process, where reaction parameters - temperature, solvent selection, pH, and crystallisation conditions - are carefully validated to ensure consistent purity and reproducibility across every batch. Our WHO-GMP certified facility in Badlapur, Maharashtra operates dedicated production streams for API synthesis, with in-house quality control laboratories equipped with HPLC, GC, IR, UV, and Karl Fischer systems for comprehensive batch testing.

Advanced infrastructure for buclizine dihydrochloride and buclizine HCl synthesis

Dedicated production lines ensuring batch-to-batch consistency

In-house quality control and analytical testing

Scalable capacity for bulk buclizine HCl supply

Reliable sourcing for pharmaceutical formulation manufacturers

Comprehensive documentation support including DMF and regulatory filings
As one of the established buclizine hydrochloride API manufacturers in India, we ensure uninterrupted supply chains and strict adherence to global quality standards.
Why Pharmaceutical Companies Source Bulk Buclizine HCl from India
Stringent regulatory environment
Indian API manufacturers operate under CDSCO oversight and are eligible for WHO-GMP, US FDA, EU GMP, and PMDA certifications, giving procurement teams confidence that quality standards meet their market requirements
Cost-competitive without quality compromise
India’s established API supply chain and skilled workforce deliver globally competitive pricing on Buclizine HCl without compromising on purity or documentation standards
Proven export infrastructure
India exports APIs to over 200 countries, with established logistics, customs expertise, and documentation experience for regulated and semi-regulated markets across Asia-Pacific, Africa, the Middle East, Latin America, and Europe
Backward integration advantage
Indian manufacturers with in-house intermediate synthesis offer supply chain resilience that import-dependent suppliers cannot, directly reducing raw material risk for your procurement team
Scale and supply continuity
India’s pharmaceutical manufacturing base is large enough to absorb demand fluctuations without compromising lead times, making it a dependable long-term sourcing region for APIs like Buclizine HCl

Bulk Supply, Packaging Options & Export Logistics
DK Pharmachem supplies Buclizine Hydrochloride API in flexible pack sizes to meet the requirements of both development-stage and commercial-scale pharmaceutical manufacturers. Standard packaging is available in 1 kg, 5 kg, and 25 kg HDPE drums with double-polyethylene inner lining to ensure protection from moisture and contamination throughout transit. Custom packing configurations are available on request.
We export Buclizine HCl API to pharmaceutical manufacturers across Asia-Pacific, the Middle East, Africa, Latin America, and Europe. Standard lead times from order confirmation are 10-15 working days for stocked material; for larger custom batches, our team will advise on specific timelines at enquiry stage. All export shipments are accompanied by a full documentation pack - Certificate of Analysis, MSDS, and any additional regulatory documents required for your destination market.
Packaging, Handling & Storage
- Supplied in secure, tamper-proof pharmaceutical-grade packaging
- Available in bulk packs as per client requirement
- Store in a cool, dry place, away from moisture and direct sunlight
- Handle with standard pharmaceutical safety protocols
- Shelf life and documentation provided as per regulatory norms
Customized packaging solutions are available for export and large-volume buyers.
For Pharmaceutical Manufacturers & Research Labs
Partner with a WHO-GMP certified supplier for consistent quality, regulatory compliance, and reliable supply of Buclizine Hydrochloride for your critical applications.
Why Choose DK Pharmachem for Buclizine Hydrochloride
Choosing the right API partner is critical for product quality and regulatory success.

Leading buclizine hydrochloride API manufacturer and exporter

Proven expertise in antiemetic APIs

Strong compliance with international quality standards

Competitive pricing for bulk buclizine HCl suppliers

Transparent documentation and technical support

Long-term reliability backed by decades of manufacturing experience
D.K. Pharmachem is trusted by pharmaceutical companies for consistent quality and dependable supply.
Frequently Asked Questions
Find answers to common questions about Buclizine Hydrochloride products and services
